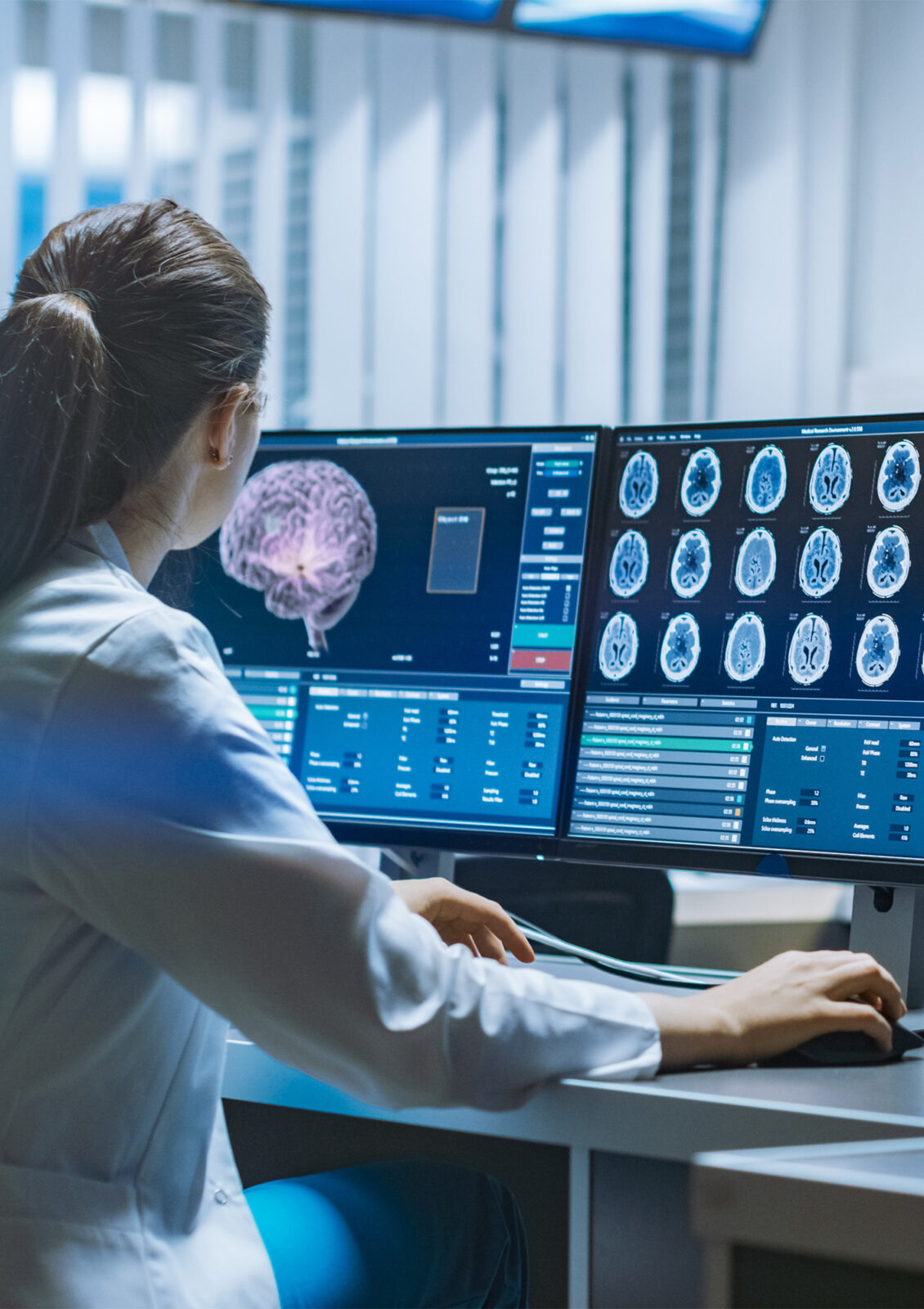
How is digitalization changing medicine?
Digitalization is changing medicine by significantly improving the efficiency and quality of patient care through the networking of medical devices and integration into existing healthcare ecosystems. Highly developed software enables more precise diagnosis, therapy planning and implementation, which has a significant positive impact on treatment outcomes.
How will digitalization influence medicine in the future?
In the future, digitalization will continue to impact medicine by driving innovative approaches and enabling the development and integration of new medical devices and technologies. These developments will not only enrich diagnosis and treatment methods, but also improve interaction and data exchange between different healthcare providers.
What role does AI play in the digitalization of medicine?
Artificial intelligence plays a crucial role in the digitalization of medicine, as it expands the possibilities of diagnosis and treatment planning through its ability to process and analyse large amounts of data. AI supports the development of more precise and effective treatment strategies, thereby helping to improve patient care. In addition, the integration of AI into medical processes promotes the research and development of new medical products and treatments.